Glucocorticoid receptors are a type of receptors on the outside of cells that transmit signals from glucocorticoids, such as cortisol. Poor glucocorticoid receptor function due to chronic stress and high CRH can lead to cortisol resistance, possibly increasing inflammation, autoimmunity, and weight gain. Read on to learn more about glucocorticoid receptors and how to optimize their function.
What Are Glucocorticoid Receptors?

Glucocorticoids are our primary stress hormones. They belong to a class of natural steroid hormones that include cortisol and cortisone. In addition, medications such as prednisone, dexamethasone, triamcinolone, and many others that end in -one are synthetic glucocorticoids and can also activate the glucocorticoid receptors [1, 2].
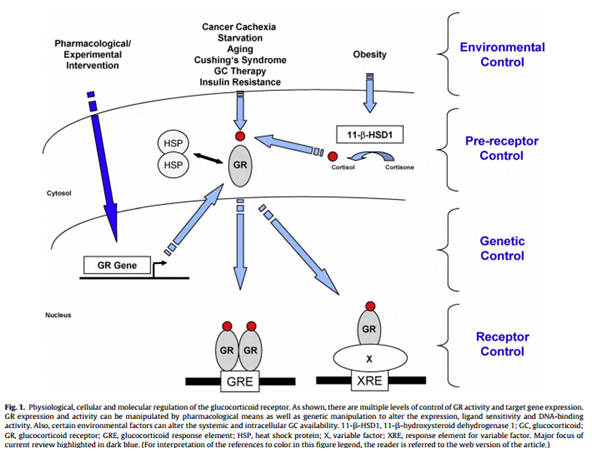
Glucocorticoid receptors are present on the surfaces of our cells. When cortisol or other glucocorticoids bind to it, the receptors move into the cell nucleus to change (increase/decrease) the production of specific proteins [1, 2].
Cortisol and the other “stress hormones” are frequently blamed for all the negative effects of stress, but this isn’t the whole story. The body’s response to cortisol is dependent on the glucocorticoid receptor, a specialized receptor that senses cortisol and related hormones [2].
While cortisol is a clear signal that stress is occurring, the glucocorticoid receptor is what dictates how the body will respond to this stress. When cortisol binds to the glucocorticoid receptor, it activates a pathway that leads to increased or decreased production of certain stress-response proteins. It’s this downstream expression, signaled by the glucocorticoid receptor, that’s really responsible for the physical effects of stress such as illness, inflammation, and anxiety [3].
Glucocorticoid (Cortisol) Resistance
Source: [4]
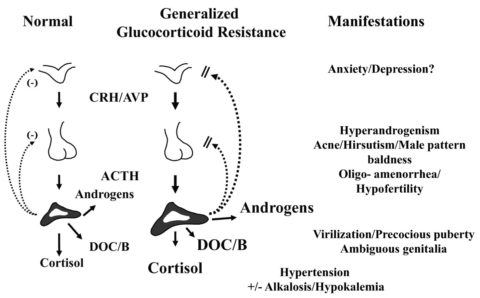
When the glucocorticoid receptor is over-activated, it becomes resistant to the effects of glucocorticoid hormones. This can cause excessive CRH and ACTH. When ACTH stimulates the adrenals, it can release other hormones like mineralocorticoids and androgens from the adrenals [4].
Causes of Glucocorticoid Receptor Resistance
1) Chronic Stress
Chronic stress is a major cause of glucocorticoid resistance. In two studies on over 350 healthy volunteers, prolonged stress caused glucocorticoid receptor activity to fall. This interferes with the appropriate regulation of inflammation and can result in more serious health risks if it is prolonged [5].
2) Steroid Medication
Long-term steroid medication use can also lead to tissue-specific glucocorticoid resistance [6, 7].
3) Inflammatory Issues
Chronic exposure to inflammatory cytokines, as in the case of inflammatory and autoimmune disorders, inhibits glucocorticoid receptor function by activating pathways (such as NF-kB, MAPK, and COX) [8].
4) Mutations in the NR3C1 Gene
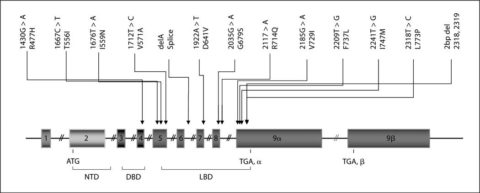
The NR3C1 gene encodes the glucocorticoid receptor. Mutations in this gene may cause glucocorticoid resistance. For instance, a mutation is responsible for a rare inherited disease called Chrousos syndrome, also known as familial or sporadic primary generalized glucocorticoid resistance [4, 9].
Location of the known mutations of the NR3C1 gene causing Chrousos syndrome
Source: [9]
Symptoms of Glucocorticoid Resistance
The following symptoms are commonly associated with glucocorticoid resistance but are insufficient to establish a diagnosis. Work with your doctor to determine the underlying condition causing your symptoms and to develop an appropriate plan to improve your health.
Glucocorticoid resistance symptoms are similar to symptoms of low cortisol. The body attempts to compensate them by increasing CRH and cortisol. CRH can cause excessive secretion of androgens and steroid hormones from the adrenals [4, 10].
Glucocorticoid resistance also causes the body to be more vulnerable to HPA axis dysfunction and inflammatory diseases, as well as to conditions like depression, fatigue, autoimmunity, and type 2 diabetes [10].
People with familial glucocorticoid resistance or those who have glucocorticoid resistance due to genetic causes suffer from some of the following symptoms [4, 11]:
- Chronic fatigue from glucocorticoid deficiency in the nervous system
- High blood pressure from excessive mineralocorticoids (since these also act on glucocorticoid receptors), which can contribute to poor cardiovascular health
- Hyperandrogenism, or signs and symptoms similar to PCOS in women, as high ACTH causes the adrenals to release high amounts of androgens (testosterone and DHEA)
- Infertility in men
- HPA axis dysfunction
Additionally, the following conditions have been associated with both familial glucocorticoid resistance and cortisol resistance from chronic stress.
1) Fatigue
Glucocorticoid resistance impairs feedback inhibition of the HPA axis, leading to its dysfunction or hyperactivity [11].
Reduced glucocorticoid receptor function also increases the production of proinflammatory cytokines. These messenger molecules may also further stimulate the HPA axis, further increasing the symptoms of the dysfunction [11].
Inflammation is one way in which cortisol resistance can cause fatigue. In addition, the lack of cortisol activity in the central nervous system can also contribute [4].
2) Depression
Depression is also strongly associated with glucocorticoid resistance in the brain, possibly because both are caused by chronic stress [12].
3) Metabolic Issues
A scientific review concluded that the dysregulation of glucocorticoid receptor activity can contribute to insulin resistance. This occurs because excessive glucocorticoid receptor activity can increase blood sugar while reducing the suppression of liver glucose production [1].
Glucocorticoid receptor signaling helps form fat cells in human tissues and can also cause abnormally high blood cholesterol or triglyceride levels (dyslipidemia) [13, 1].
Increased activity of the glucocorticoid receptor in the muscles is associated with metabolic syndrome (a group of conditions that increase the risk of heart disease, stroke, and diabetes) [2].
Glucocorticoid hypersensitivity is associated with insulin resistance and high blood pressure, which are potentially serious health concerns by themselves in addition to being risk factors for type 2 diabetes [14].
Because mutations in the NR3C1 gene impair glucocorticoid receptor action and alter tissue sensitivity to glucocorticoids, they are strong risk factors for these issues [9].
4) Infertility and PCOS
Women with familial glucocorticoid resistance frequently experience acne, facial hair, menstrual irregularities, problems with ovulation, and infertility [4].
Many women with polycystic ovarian syndrome (PCOS) also have cortisol resistance [15].
Glucocorticoid resistance is also associated with infertility in men [4].
5) Immune Responses
Cortisol is an anti-inflammatory hormone that can deactivate immune responses. Stress can induce immune cells, such as macrophages, to become resistant to cortisol [16].
Chronic stress can weaken the immune system and make you more susceptible to a cold. In addition, people who develop colds amidst stress have worse symptoms because their immune cells are resistant to inhibition by cortisol [5].
Conditions Associated with Excess Activation
The following conditions have been associated with excessive glucocorticoid receptor activation. Note, however, that most studies were associational, meaning a cause-and-effect relationship hasn’t been established. Because the conditions are complex, other genetic and environmental factors could have contributed.
1) Cancer
Glucocorticoid receptors play a role in increasing cell survival and genes that stop programmed cell death in breast cancer patients. A high glucocorticoid receptor production was associated with a poor prognosis in breast cancer patients in a study on almost 1,400 women [17, 18].
Glucocorticoid receptor activation prevented tumor growth in prostate cancer cells [17].
Glucocorticoid and androgen receptors share several transcriptional targets. Androgen receptor (AR) signaling can increase tumor formation in prostate cancer. AR-targeted therapy is used to help treat prostate cancer by blocking the androgen receptor function [17].
When androgen receptors are inhibited in cell culture, glucocorticoid receptors’ expression increases. The increase in glucocorticoid receptors, in turn, promotes the expression of the shared target genes. These shared target genes include AR-regulated survival-promoting genes, which increase tumor cell survival [17].
2) Weakened Bones and Muscles
Animal studies found that high glucocorticoid receptor activity promoted bone loss and osteoporosis [2].
Researchers also concluded that glucocorticoid receptor signaling may cause muscle wasting in animals, suggesting that high glucocorticoid receptor signaling could be a risk factor for this condition [2].
3) Alcohol Dependence
In alcohol-dependent rats, glucocorticoid receptor signaling in the central amygdala contributed to compulsive drinking. After exposure to alcohol, the rats’ glucocorticoid receptor activity increased and lead to HPA dysfunction [19].
Mifepristone is a drug that blocks glucocorticoid receptor activity. In alcohol-dependent patients, treatment with Mifepristone reduced alcohol-cued craving and drinking. These results suggest that glucocorticoid receptor activity plays a role in alcoholism [19].
Benefits
The stress hormones are mostly discussed for their negative effects. However, healthy glucocorticoid receptor function and signaling can reduce inflammation, improve mood, and increase energy levels. Some of the main benefits of healthy receptor activation are discussed below.
1) Reducing Inflammation
Glucocorticoids can reduce inflammation through their interactions with the glucocorticoid receptor.
When activated, the glucocorticoids receptor can increase proteins that limit inflammation. This activation can also increase the activity of the beneficial antioxidant enzyme superoxide dismutase [20].
Activation of the glucocorticoid receptors can also trigger a decrease in the production of inflammatory factors. Glucocorticoid receptors reduce inflammation by limiting the activity of the inflammatory proteins AP-1 and NF-κB [20, 21].
These effects are not straightforward and can differ widely from different causes and levels of inflammation. Current evidence suggests that glucocorticoid receptor activation may not be effective in reducing inflammation in more serious immune disorders [10].
2) Improving Mitochondrial Health
Many steroid and thyroid hormone receptors increase mitochondrial function to protect cells. Glucocorticoid receptors (GR) form a complex with the Bcl-2 protein to control brain mitochondria [22].
In animal brain cell cultures treated with corticosterone (the animal equivalent of cortisol), a GR/Bcl-2 complex is formed. The complex then moves into the mitochondria and helps prevent brain cell degeneration [22].
3) Preventing Depression
Decreased glucocorticoid receptor function also contributes to HPA axis hyperactivity and to the development of depressive symptoms [23].
To keep these diseases and behavioral alterations from happening, properly functioning glucocorticoid receptors maintain the body at homeostasis. In other words, the feedback from the glucocorticoid hormones should help prevent depression in healthy people [24].
Depression is also strongly associated with glucocorticoid resistance, possibly because depression and glucocorticoid resistance are both caused by chronic stress [12].
Indeed, reversing glucocorticoid resistance is one of the ways in which many antidepressants work [25].
4) Reducing Autoimmune Disease
Glucocorticoid receptors increase anti-inflammatory proteins and help prevent inflammation, which protects the immune system [20].
Glucocorticoid receptors in T cells within the intestine play a role in controlling immune activation. Peripheral T cells also need these receptors for their immunosuppressive effects [26].
5) Suppressing Skin Cancer
In skin cancer, glucocorticoid hormones can cause tumor growth inhibition, induction of cancer cell death, and inhibition of tumor cell motility, as seen in a cell-based study [21].
Glucocorticoid receptors use the same mechanism when inhibiting pro-inflammatory genes to prevent tumor cell growth. Glucocorticoid receptor signaling inhibits cyclin-dependent kinase (Cdk4), which plays an important role in skin cancer [21].
In mice, glucocorticoid receptor signaling also lowered the number of follicular epithelial cells, which are an important target cell population for skin tumor formation [21].
6) Protecting Hearing
The glucocorticoid receptors may also help to protect the body against hearing loss associated with loud noises. Animal studies showed that glucocorticoid receptors are activated in the ear during auditory stress. This decreases the auditory brainstem threshold, which reduces the hearing loss caused by trauma [27].
Factors That Increase Glucocorticoid Receptor Function
1) Drugs
- Dexamethasone (a synthetic glucocorticoid/steroid medication) has a high affinity for glucocorticoid receptors [26].
- Selective glucocorticoid receptor agonists and modulators (SEGRAM) are experimental drugs that trigger certain glucocorticoid receptor mechanisms. However, they are still in development and not much information is available [28].
- Antidepressants, such as clomipramine, amitriptyline, and paroxetine, enhanced glucocorticoid receptor function in cell-based studies, especially in combination with dexamethasone [25].
2) Stress Management
Chronic stress can cause glucocorticoid resistance. Read this post to learn about SelfHacked’s stress management strategies.
3) Others
- In mice white blood cell (macrophages), the cytokine IFN-gamma increased glucocorticoid receptors [29]
- Ginsenosides (from ginseng) increased the binding activities of glucocorticoid receptors in mice brain, lung, and liver cells [30]
- Chronic lithium chloride injection increased glucocorticoid receptor production (mRNA expression) in rat brains [31]
Factors That Decrease Glucocorticoid Receptor Activity
1) Natural Substances
- High vitamin B6 concentration suppressed glucocorticoid receptor production, while its deficiency enhanced the body’s responsiveness to glucocorticoids, in rats and cells [32].
- Curcumin inhibited the glucocorticoid receptor’s ability to change cellular production in a study in human cells [33].
- Luteolin stopped both glucocorticoid and progesterone signaling in human cells [34].
2) Drugs
- Mifepristone, a drug used for medical abortions and emergency contraception, lowered glucocorticoid receptor activity in cells [35].
- Ketoconazole and related drugs that treat fungal infections inhibit glucocorticoid receptor transcriptional activity in cells [36].
- RU-43044 specifically inhibited glucocorticoid receptor activity in depressed mice [37].
3) Others
Certain cytokines can decrease glucocorticoid receptor production. These include IL-1 alpha, IL-2, IL-13, IFN-alpha, TNF-alpha, and IL-6 [38].
The glucocorticoid receptor and the inflammatory molecule NF-κB can inhibit each other’s activity [38, 39].
In cells isolated from asthmatic patients, exposure to allergens decreased glucocorticoid receptor sensitivity. Ragweed pollen and cat hair also caused the cells to be less sensitive to glucocorticoids (such as hydrocortisone and dexamethasone) [40].
Toxins such as aflatoxin, anthrax, and arsenic decreased glucocorticoid receptor activity in cells [41].
RNA growth arrest-specific 5 (Gas5) builds up in cells to repress glucocorticoid receptors. Its levels are mostly higher in blood cells of patients with autoimmune and infectious diseases [42].
Gas5 decreases glucocorticoid receptor activity with its decoy RNA glucocorticoid receptor elements (GREs). Changes in Gas5 levels influence how much of a therapeutic effect glucocorticoid receptors can have on diseases [42].
The Cannabinoid System
The endocannabinoid system is a fat signaling system of the brain and body. In addition to cannabinoids produced by the body (such as anandamide), the system is activated by the cannabinoids from marijuana THC and CBD. The system has two receptors, CB1 and CB2, but most research is focused on the CB1 receptor [43].
Under normal conditions, endocannabinoid signaling decreases HPA axis activation. When CB1 receptor signaling is disrupted, it increases HPA axis activity [43].
The endocannabinoid system contributes to both the short- and long-feedback loops of glucocorticoid feedback inhibition. Glucocorticoids inhibit the HPA axis by stimulating the endocannabinoid activity in the hypothalamus, amygdala, and hippocampus [43, 44].
Prolonged glucocorticoid administration can decrease CB1 receptor density in the hippocampus without affecting endocannabinoid levels [45].
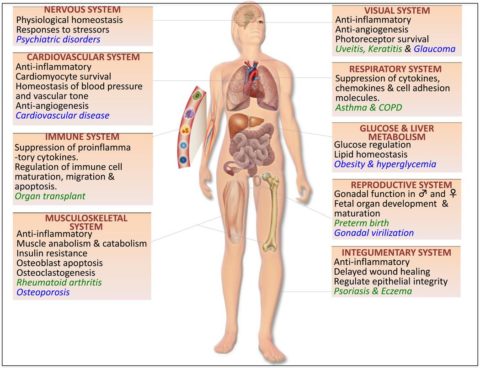
Mechanisms
The roles of glucocorticoids in major organ systems of the human body (black text), beneficial roles of glucocorticoids in the clinic (green text), and adverse outcomes in patients with elevated glucocorticoid levels (blue text).
Source: [2]
- The PPARGC1B (PGC-1beta) gene increases glucocorticoid receptor activity in the presence of glucocorticoids.
- NCOA2 is a coactivator of NR3C1 and increases its transcriptional activity by working with RWDD3.
- Glucocorticoid receptor activation inhibits mitogen-activated protein kinases (MAPK) via direct negative interaction on a protein-protein level or inhibition of MAPK phosphorylation. MAPKs are key mediators of the cytokine-induced inflammatory response. The negative cross-talk between GR- and MAPK prevents inflammation [21].
- Glucocorticoid receptor activation may increase the transcription of genes coding for anti-inflammatory proteins, including lipocortin-1, interleukin-10, and interleukin-1 receptor antagonist [20].
- Preliminary evidence in mice involved glucocorticoid receptor signaling in maintaining glucose homeostasis [2].
- Excess glucocorticoid receptor activity may also cause systemic dyslipidemia by increasing the expression and secretion of ANGPTL, leading to excessive inhibition of lipoprotein lipase (LPL) activity in fat tissue [1].
- Recurrent miscarriages have been associated with certain polymorphisms in NR3C1. However, other studies failed to confirm these results [46].
Genetics
Glucocorticoid Receptor Polymorphisms
The NR3C1 (or GCR) gene encodes the glucocorticoid receptor [2].
The main polymorphisms of the NR3C1 gene are BclI, N363S, ER22/23EK, and GR-9β [2].
- BclI (rs41423247) is associated with obesity, depression, and hypersensitivity to glucocorticoid treatment.
- N363S (rs6195) is associated with improved glucocorticoid sensitivity.
- ER22/23EK (rs6189 and rs6190) is associated with decreased glucocorticoid sensitivity and increased BMI, triglyceride and cholesterol levels, muscle mass, and risk of depression.
- GR-9β (rs6198) is associated with increased glucocorticoid receptor resistance, and risk for rheumatoid arthritis and heart disease or heart attacks.
However, since glucocorticoid receptor polymorphisms are rare, the associations between the polymorphisms and risk for certain diseases may not be significant [2].
Glucocorticoid-Responsive Elements and Gene Expression
Target DNA sequences are called glucocorticoid-responsive elements (GREs). GREs bind to the glucocorticoid receptor to express target genes [47].
DNA-binding proteins control the sensitivity of certain GREs. Some GREs have very low concentrations of glucocorticoids, so they are considered to be hypersensitive. On the other hand, some GREs need high doses for glucocorticoid receptor binding [47].
Glucocorticoid-activated receptors activate gene expression in three primary ways. The receptors can bind directly to DNA, tether to other DNA-bound transcription factors, or interact with nearby DNA-bound transcription factors [47].
Glucocorticoid receptors can also bind to negative GREs, which suppresses the transcription of some genes [48].
Genes That Glucocorticoid Receptors Regulate
Glucocorticoid Receptor Activation Increases the Expression of:
- GILZ (glucocorticoid-induced leucine zipper) [49]
- SGK1 (serum/glucocorticoid-regulated kinase 1) [50]
- TTP (tristetraprolin) [51]
- MKP-1 (mitogen-activated protein kinase phosphatase-1) [52]